|
More attention than ever is being put on your gut health, and understandably so, considering a significant proportion of your immune system resides in your gastrointestinal tract.1 As such, optimizing your gut microbiome is a worthwhile pursuit that will have far-reaching effects on your physical health and emotional well-being. Mounting scientific evidence also continues to suggest a large component of nutrition centers on nourishing health-promoting bacteria in your gut (and elsewhere in and on your body). In doing so, you keep harmful microbes in check and shore up your protection against chronic disease. Disease Begins in Your GutADHD, autism, learning disabilities, obesity, diabetes2 and Parkinson’s disease are but a few of the conditions found to be influenced by your gut microbiome. One 2020 scientific review3 goes so far as to say that all inflammatory disease begins in the gut. Part of the blame is laid on excessive hygiene. In other words, we’re “too clean” for our own good. But your diet also plays a crucial role. The paper specifically addresses the role of zonulin-mediated gut permeability in the pathogenesis of chronic inflammatory diseases (CIDs). According to the author, Dr. Alessio Fasano,4 a pediatric gastroenterologist, researcher and director of the Center for Celiac Research and Treatment:5
Bacteria, Not Genes, Rule Your Health DestinyFasano points out that we simply do not have enough genes to account for the myriad chronic diseases that can beset us. Genes also cannot explain the timing of disease onset. To solve these mysteries, we must look to the microbiome, he says, as “it is the interplay between us as individuals and the environment in which we live that dictates our clinical destiny.” Aside from the microbes themselves, the condition of your intestinal mucosa also plays a significant role. “Although this enormous mucosal interface (200 m2) is not apparently visible, it plays a pivotal role through its dynamic interactions with a variety of factors coming from our surrounding environment, including microorganisms, nutrients, pollutants and other materials,” Fasano explains. While intracellular tight junctions used to be thought of as static and impermeable, we now know this is not the case. As explained by Fasano, zonulin is a powerful modulator of intestinal permeability. However, while zonulin is a biomarker of gut permeability and plays a pathogenic role in in many chronic inflammatory diseases, not all CIDs are caused by leaky gut. Proposed Chain of Events Leading to CIDThe graphic below, included in Fasano’s review but originating from an earlier paper6 titled “Zonulin, a Regulator of Epithelial and Endothelial Barrier Functions, and Its Involvement in Chronic Inflammatory Diseases,” co-written by Fasano and Craig Sturgeon, details the “proposed chain of events leading to chronic inflammatory disease.” 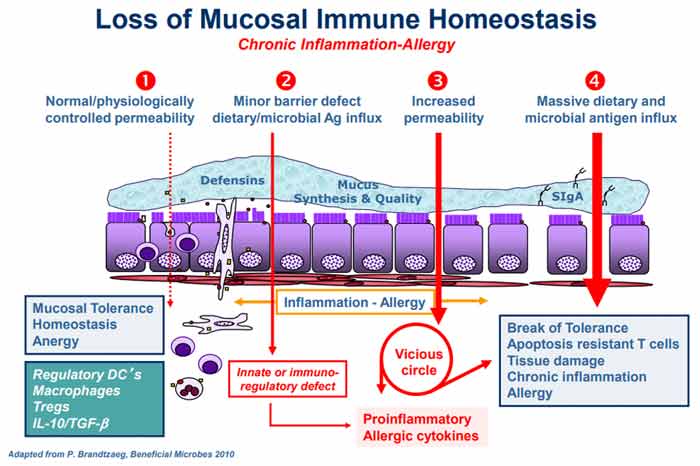
Under normal circumstances, a healthy homeostasis is maintained in your gut lining such that when an antigen is encountered, no excess immune reaction occurs (anergy). Under No. 2 in the graph, gut dysbiosis is setting in (i.e., an imbalance in the number and diversity of your gut microflora), causing excess production of zonulin, which in turn makes the gut lining more permeable. According to Fasano, the two most powerful triggers of zonulin release are bacteria overgrowth and gluten. Zonulin is produced in response to bad bacteria7 — it helps flush the bacteria out by opening up the tight junctions — so bacteria overgrowth makes sense. But why does it respond to gluten? Interestingly enough, the zonulin pathway misinterprets gluten as a potential harmful component of a microorganism. That’s why gluten triggers zonulin release. While not mentioned by Fasano, the herbicide glyphosate also triggers zonulin, and is 10 times more potent than gluten!8 The subsequent permeability allows microbiota-derived antigen and endotoxin to migrate from the lumen to the lamina propria (the connective tissue that is part of the mucous membrane lining your intestine), thereby triggering inflammation. As the process continues to worsen (No. 3 in the graph), your adaptive immune response kicks in, triggering the production of proinflammatory cytokines, including interferon gamma (IFN-γ) and tumor necrosis factor alpha (TNF-α). These cytokines further worsen the permeability, thus creating a vicious cycle. Eventually (No. 4), mucosal tolerance is completely broken, resulting in the onset of a chronic inflammatory disease. Chronic Inflammatory Diseases Linked to Leaky GutThe specific chronic inflammatory disease that ultimately emerges at the end of all this depends in part on your genetic makeup, in part on the types of exposures you’ve had, and in part on the composition of your gut microbiome. As explained by Fasano:9
Chronic inflammatory diseases associated with dysregulation of the zonulin pathway include:
Gut Microbes Influence Genes and Can Influence Cancer RiskWhile the inclusion of cancer on that list may seem odd at first glance, some researchers believe the gut microbiome may actually end up being a game-changer for cancer prevention and treatment. Not only have gut bacteria been shown to influence gene expression,10,11 turning some genes on and others off, research12 published in 2018 found gut microbes actually control antitumor immune responses in your liver, and that antibiotics can alter the composition of immune cells in your liver, triggering tumor growth. Certain gut bacteria also promote inflammation, which is an underlying factor in virtually all cancers, whereas other bacteria quell it.13 The presence of certain gut bacteria has even been shown to boost the patient’s response to anticancer drugs.14 One way in which gut bacteria improve the effectiveness of cancer treatment is by activating your immune system and allowing it to function more efficiently. Researchers have actually found that when these specific microbes are absent, certain anticancer drugs may not work at all. Gut Bacteria Are Part of Your Antiviral DefenseGut bacteria are also involved in your antiviral defense, recent research15 shows. As reported by Harvard Medical School November 18, 2020:16
While the findings still need to be replicated and confirmed, they point to the possibility that you might be able to enhance your antiviral immunity by reseeding your gut with Bacteroides fragilis and other bacteria in the Bacteroides family.17 These bacteria initiate a signaling cascade that induces the release of interferon-beta that protect against viral invasion by stimulating immune cells to attack the virus and causing virus-infected cells to self-destruct.
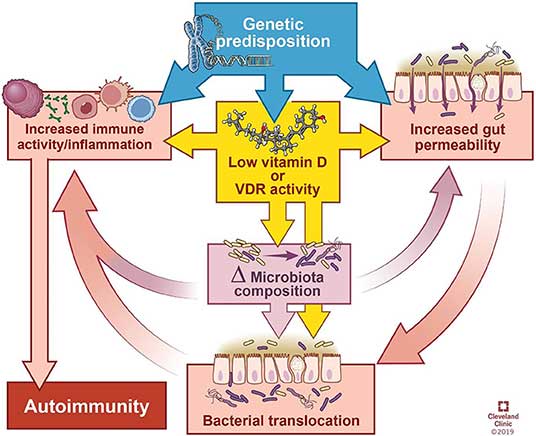
The Role of Vitamin DRecent research also highlights the role of vitamin D in gut health and systemic autoimmunity. The review article, published January 21, 2020, in Frontiers in Immunology, notes:19
As noted in this review, vitamin D has several direct and indirect regulatory effects on your immune system, including promoting regulatory T cells (Tregs), inhibiting differentiation of Th1 and Th17 cells, impairing development and function of B cells, reducing monocyte activation and stimulating antimicrobial peptides from immune cells. That said, the relationship between vitamin D and autoimmunity is complicated. Aside from immunosuppression, vitamin D also appears to improve autoimmune disorders by the way it affects your microbiota composition and gut barrier. The review cites research showing that your vitamin D status alters the composition of your gut microbiome. Generally speaking, vitamin D deficiency tends to increase Bacteriodetes and Proteobacteria while higher vitamin D intake tends to increase prevalence of Prevotella and reduce certain types of Proteobacteria and Firmicutes. While research is still slim when it comes to vitamin D’s impact on gut bacteria, especially in patients with autoimmune disease, vitamin D deficiency and autoimmune diseases are known comorbidities and vitamin D supplementation is often recommended for these patients. Vitamin D Required for Tight Junction MaintenanceBetter known is how vitamin D supports intestinal and immune cell defenses in the gut. In fact, vitamin D is one of the crucial components required for maintaining tight junctions. As explained in this review:20
How Vitamin D May Contribute to Autoimmune DiseaseAccording to the authors, vitamin D deficiency may contribute to autoimmune disease by affecting the microbiome and the immune system in the following manner:
How to Optimize Your Gut MicrobiomeAll of this information should really drive home the point that optimizing your gut flora and vitamin D level is of crucial importance for good health. By reseeding your gut with beneficial bacteria, you can keep pathogenic microbes and fungi in check and prevent them from taking over, and optimizing your vitamin D will help avoid leaky gut. Regularly eating traditionally fermented and cultured foods is the easiest, most effective and least expensive way to make a significant impact on your gut microbiome. Healthy choices include lassi (an Indian yogurt drink), cultured grass fed organic milk products such as kefir and yogurt, natto (fermented soy) and fermented vegetables of all kinds. Although I'm not a major proponent of taking many supplements (as I believe the majority of your nutrients need to come from food), probiotics are an exception if you don’t eat fermented foods on a regular basis. Spore-based probiotics, or sporebiotics, can be particularly helpful when you’re taking antibiotics. They’re also an excellent complement to regular probiotics. Sporebiotics, which consist of the cell wall of bacillus spores, will help boost your immune tolerance, and because they do not contain any live bacillus strains, only its spores — the protective shell around the DNA and the working mechanism of that DNA — they are unaffected by antibiotics. Antibiotics, as you may know, indiscriminately kill your gut bacteria, both good and bad. This is why secondary infections and lowered immune function are common side effects of taking antibiotics. Chronic low-dose exposure to antibiotics through your food also takes a toll on your gut microbiome, which can result in chronic ill health and increased risk of drug resistance. Last but not least, you also need to avoid things that disrupt or kill your microbiome, and this includes:
from http://articles.mercola.com/sites/articles/archive/2020/12/31/the-gut-microbiome-in-health-and-disease.aspx
0 Comments
Leave a Reply. |
Nia Pure NatureThe Provider of premium Quality Health Products To Live Better Lives Archives
March 2022
Categories |

 RSS Feed
RSS Feed
